Background
Since 2002, SIAPS and its predecessor programs, SPS and RPM Plus, have supported Amazon Malaria Initiative (AMI) countries to have institutionalized and sustainable national and regional mechanisms that will ensure a continuous supply of antimalarial medicines as the key malaria control strategy, particularly in low-incidence areas and remote areas where underserved populations live and work. The SIAPS AMI Project worked in collaboration with the Pan American Health Organization (PAHO) Infectious Disease Division, the US Centers for Disease Control and Prevention, the US Pharmacopeia’s Promoting Quality of Medicines Program, the national malaria control programs in the Amazon and Central American regions, and USAID missions.
Over the last decade, there has been a decline in the number of malaria cases in the Amazon Basin due to the introduction of artemisinin-based combination therapy (ACT), which has strengthened malaria control strategies. A regional analysis of antimalarial supply chain management highlighted the main challenges in ensuring uninterrupted access to antimalarials in low-incidence areas. These challenges included limited commercial interest of pharmaceutical suppliers to participate in national tenders, particularly for medicines with a low volume of consumption such as those used to treat severe and atypical malaria cases. SIAPS supported baseline studies and interventions to improve the availability of antimalarials in decentralized, high-incidence areas of Colombia, including Chocó, which has the second highest malaria burden in the country.
Project Highlights
SIAPS supported the design and implementation of procedures for product distribution, warehousing improvement, and implementation of malaria pharmaceutical guidelines for primary health care facilities. SIAPS also disseminated a bottleneck analysis to improve adherence to the PAHO/Strategic Fund (SF) pooled procurement and transferred the coordination of the antimalarial regional stock monitoring system to the SF.
Results
SIAPS/AMI has made significant strides toward institutionalizing strategies and activities to ensure a continuous supply of antimalarials in low-incidence areas and areas prone to ACT resistance.
- Guidelines and procedures:
- Colombia developed a guideline entitled What I Need to Know about Antimalarial Supply Management at the Primary Level of Care: Colombia–2013. It is intended to promote the guide to other areas of the country where malaria is endemic.
- SIAPS developed guidelines and standard operating procedures for distributing and using malaria medicines in Colombia. The Vector-Borne Disease Program of the Health Secretariats helped implement the guide.
- Capacity building:
- Work sessions were conducted with staff in the technical, communication, and operations management units of Ministry of Health and Social Protection (Ministerio de Salud y Protección).
- Medicine availability: Based on the revised estimation of need and considering security stock for low-incidence areas, the Regional Stock Monitoring System for Antimalarial has contributed to an increased availability of medicines across the region. The AMI central medicine stores have, on average, 85% of malaria medicines available. A quarterly bulletin that outlines levels of antimalarial stock has facilitated the requisition and donation of first-line antimalarials among countries and the donation of medicines for severe malaria through the PAHO SF.
[Click to enlarge]
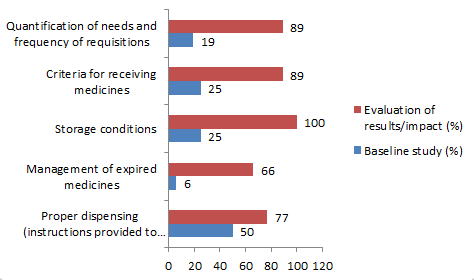
In Chocó, increased staff knowledge of critical areas of the supply process contributed to the improved availability of medicines. There was marked improvement in storage conditions (from 25% to 100%), criteria for receiving medicines (from 25% to 89%), and quantification of needs and frequency of requisitions (from 19% to 89%).
References
- Hinestroza Y. 2012. Evaluación de la gestión del suministro de medicamentos antimaláricos e insumos para diagnóstico de la malaria. Departamento del Chocó-Colombia-2012 – Línea de base. Presentado a la Agencia de los Estados Unidos para el Desarrollo International por el Programa Systems for Improved Access to Pharmaceuticals and Services (SIAPS). Arlington, VA: Management Sciences for Health
- 2015. Informe técnico: Evaluación de la situación de la gestión del suministro de medicamentos antimaláricos en los países miembros de la Iniciativa para el Control de la Malaria en la Cuenca del Amazonas. Presentado a la Agencia de los Estados Unidos para el Desarrollo Internacional por el Programa Systems for Improved Access to Pharmaceuticals and Services (SIAPS). Arlington, VA: Management Sciences for Health
- What I need to know about Antimalarial Supply Management at the Primary Level of Care: Colombia—2013
- Other documents

