By Helena Walkowiak, Principal Technical Advisor, SIAPS
It’s been called the pharmaceutical sector, a drug supply or management system or the medical products building block. By any name, the part of a health system that deals with ensuring access to essential medicines, vaccines and medical products and their correct use–we call it a pharmaceutical system at SIAPS–is critical. Yet despite much research in the field on improving its performance and considerable progress towards strengthening its components, there is no apparent consensus on what constitutes a pharmaceutical system in all of its complexity.
In addition, there’s no clearly defined framework for measuring progress in pharmaceutical system strengthening (PSS). That means that countries and donors lack complete information for guiding their investments in PSS interventions, and the tools and agreed-on measures to evaluate them.
We can and do measure progress in improving individual components of a pharmaceutical system; for example, in medicines procurement or regulatory systems. But without a blueprint for the entire system, we might miss critical aspects that are weak – and that could undermine the efforts of countries to improve access to and proper use of needed medicines. These weaknesses may also bring the system to a grinding halt over time or compromise its ability to handle change or disruptions.
In a paper just published in Health Policy and Planning, I and my co-authors have proposed definitions for a pharmaceutical system and PSS, plus the components and attributes critical for measuring it.
I recently summarized our findings and our proposed definitions and concepts at the Fourth Global Symposium on Health Systems Research conference in Vancouver. To develop them, we carried out a comprehensive literature review and looked at existing definitions, frameworks, and assessment tools. Our review revealed the wealth of information but also the lack of consensus that exists. SIAPS then convened a meeting of partners and experts to discuss the findings and we worked together to reach agreement.
So what are a pharmaceutical system and pharmaceutical systems strengthening?
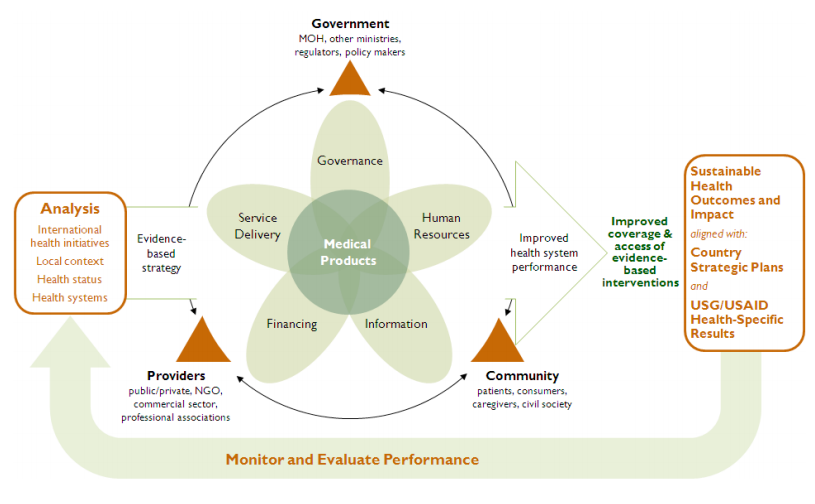
Our definition proposes that a pharmaceutical system consists of:
All structures, people, resources, processes, and their interactions within the broader health system that aim to ensure equitable and timely access to safe, effective, quality pharmaceutical products and related services that promote their appropriate and cost-effective use to improve health outcomes.
We further propose that pharmaceutical systems strengthening is:
The process of identifying and implementing strategies and actions that achieve coordinated and sustainable improvements in the critical components of a pharmaceutical system to make it more responsive and resilient and to enhance its performance for achieving better health outcomes.
Resilience is a key characteristic of a well-functioning pharmaceutical system, so the concept of resilience is embedded in our definition. We also refer to improvements as “sustainable” to take into account the distinction between strengthening interventions versus those that support the system and may depend on continued external inputs to sustain improved performance.
Now that we have defined PSS, how do we measure it?
We identified seven components of PSS measurements:
- Pharmaceutical products and related services
- Policy, laws and governance
- Regulatory systems
- Innovation, research and development, manufacturing, and trade
- Financing
- Human resources
- Information
We also included two attributes of the pharmaceutical system—performance and resilience—that provide a practical starting point for assessing progress towards stronger and more resilient pharmaceutical systems.
Measuring outcomes will also need to track the primary outcomes of a pharmaceutical system, namely access — affordability, acceptability, accessibility and availability – to safe, quality, and effective products and their appropriate, cost effective use.
We recognize that this is simplified way at looking at what is a very complex network and its relationship with the broader health system, but it’s also one that lends itself to the development of practical tools that countries and their partners can deploy to measure and monitor results. Given the importance of pharmaceutical systems to achieving the health and risk protection goals of universal health care, our work completes an important step towards evaluating PSS interventions and investments.
Source: Hafner, Tamara, Helena Walkowiak, David Lee, and Francis Aboagye-Nyame. “Defining pharmaceutical systems strengthening: concepts to enable measurement.” Health Policy and Planning, 2016. doi:10.1093/heapol/czw153
Blog includes contributions from Stacy Lu, SIAPS Technical Writer

