The lack of a trained health care workforce creates a barrier to the delivery of quality health services. Inadequate knowledge and limited or outdated skills can be obstacles to achieving positive health outcomes. Advances in the treatment and diagnosis of diseases, new technologies, and systems and process improvements, as well as changes in professional roles and responsibilities, require health care workers to engage in continuing professional development (CPD). To address both changing professional roles and advancements in patient management, a number of organizations and donors have created continuing education (CE) and training activities for pharmaceutical management in resource-limited countries. The challenge is in coordinating these CE activities and ensuring quality CE is delivered in resource-limited settings.
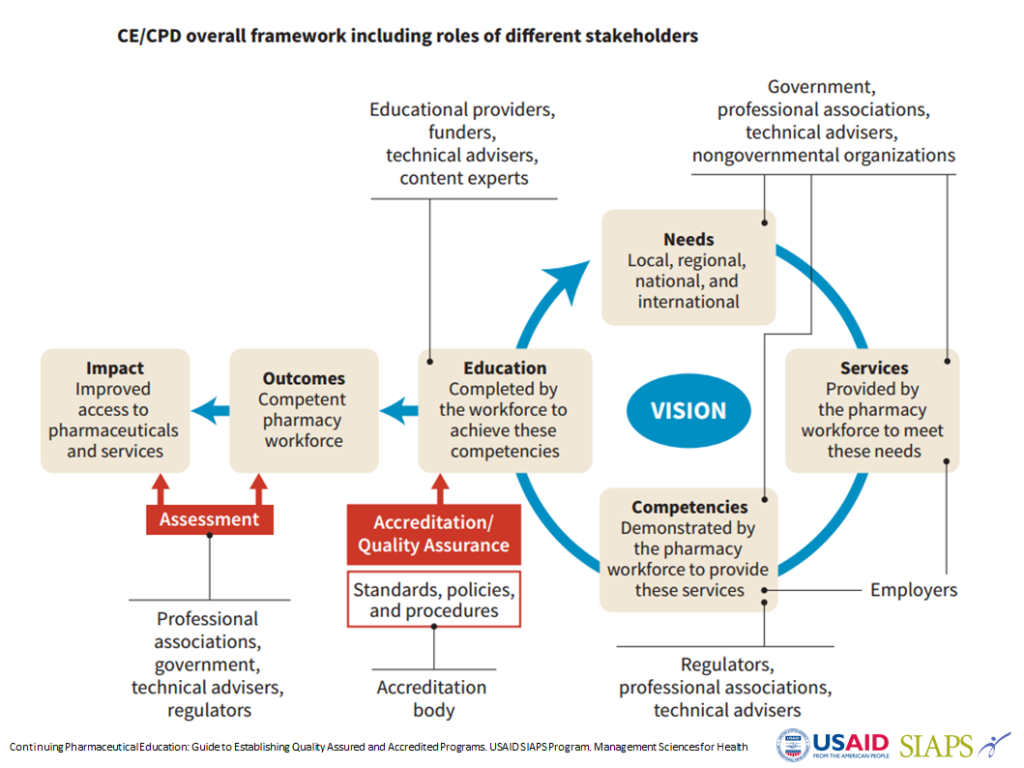
CPD is essential to the maintenance and advancement of health care worker competency. For example, without CPD, pharmacy support professionals may not know how to store new family planning devices to ensure their quality, or prescribers may misinterpret their country’s policy on Option B+ for antiretroviral treatment. Pharmaceutical professionals, including pharmacists and pharmacy support personnel, can enhance their knowledge and skills by engaging in CPD through quality CE and training activities. Professional agencies can ensure that pharmaceutical personnel have the requisite skills by utilizing the quality assurance criteria and benchmarking standards for individual activities that help ensure the quality of CE/CPD and training activities. These quality criteria and standards should address the “Pillars of Educational Quality” (context, structure, process, outcomes, and impact) and rest on the “Foundations” (science, practice, and ethics) of quality education. What has been lacking is a quality assurance framework for guiding the delivery of CPD/CE for pharmacy professionals in low-resource settings.
To meet this need, the Systems for Improved Access to Pharmaceuticals and Services Program developed “Continuing Pharmaceutical Education: Guide to Establishing Quality Assured and Accredited Programs” in partnership with the Accreditation Council for Pharmacy Education. This guidance document and its associated tools were designed to ensure the quality of CPD activities and advance educational excellence for the pharmacy profession by establishing accreditation standards and criteria for CPD and CE programs. This document guides the establishment and benchmarking of accreditation programs for continuing pharmaceutical education and training in low- and middle-income countries. It is a resource for governments and other stakeholders for establishing and maintaining high-quality, accredited programs for pharmaceutical training, CE, and CPD activities to ensure that the knowledge and skills-based training offered to pharmacy staff meet a defined quality standard.
Countries can use this document to guide the development of an accreditation process that will ensure the quality of CE/CPD activities and, as a result, help to improve pharmaceutical services. Although this guidance focuses on CE for pharmacy professionals, the principles and approach are also applicable to CE for other health care workers, including medical doctors, nurses, and allied health professionals. The document also guides quality improvement initiatives for countries with existing CE/CPD accreditation procedures. Because countries may be at different stages in the implementation of quality assurance programs for CE activities, the guidance can be applied either in whole or in part and is compatible with the health systems strengthening priorities of sub-sector specific (e.g., CPD for pharmaceutical personnel in the faith-based sector) or national programs.

