Improving Medicines Access and Use for Child Health—A Guide to Developing Interventions represents an up-to-date[i] and practical resource for those developing interventions to improve access to and use of medicines for child illness. While it primarily targets groups working in community organizations, health facilities, and district health offices, it could also be used within the larger health system at national level.
How is the Guide Organized?
The guide adopts a structured approach to lead a team through five steps (see Figure 1). Each step is addressed in a separate chapter, with linked annexes containing detailed tables, resource lists, supporting materials, and case examples. Teams that have already identified and explored problems can skip earlier chapters.
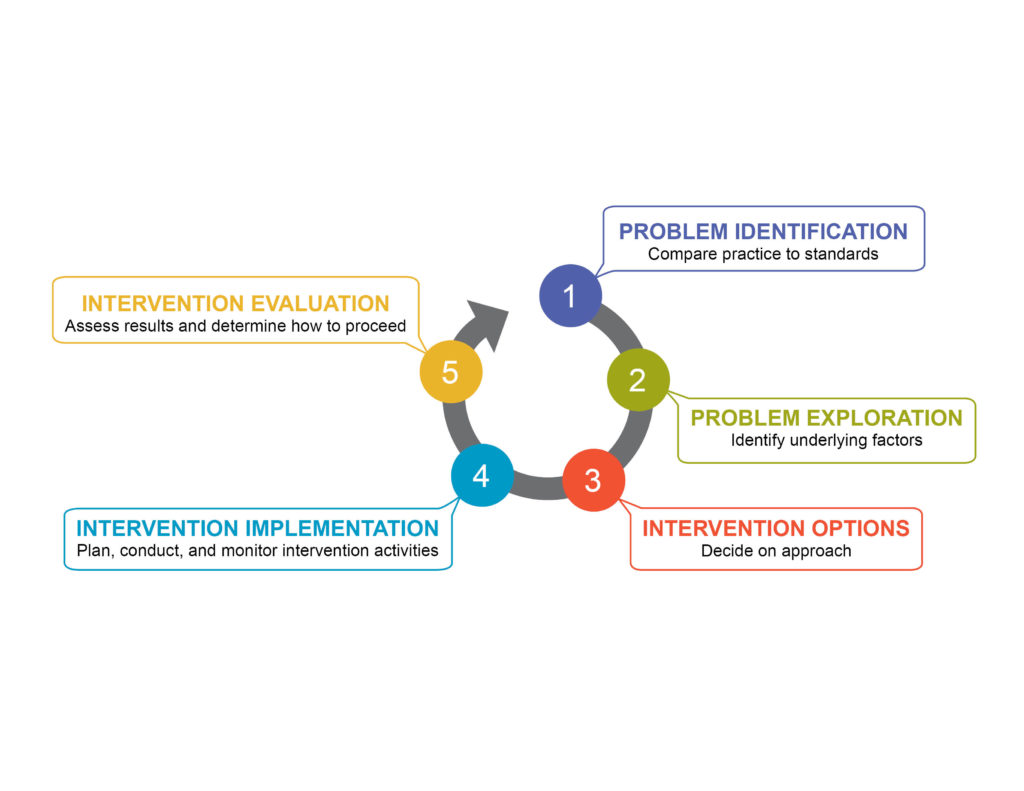
- Chapter 1 introduces a framework for identifying problems that interfere with the effective use of medicines to treat sick children. It describes tools for assessing caregiver and provider practices, using appropriate indicators at each step of the sick child care pathway.
- Chapter 2 discusses methods for exploring the common causes of problems and difficulties related to access and use of medicines for children.
- Chapter 3 presents options to address complex issues with practical solutions. It orients the user in the selection of various interventions to improve access and use of medicines targeting caregivers, health providers, or the care system as a whole.
- Chapter 4 orients users in the design, implementation, and monitoring of interventions by reviewing some of the principles for mounting effective multifaceted interventions and describes how intervention components can reinforce each other.
- Chapter 5 offers guidance on how to evaluate the results of an intervention in a particular setting to strengthen its impact and disseminate successful strategies.
Who Should Use the Guide?
This guide will be useful to policy makers and health system managers working in the public, mission, or nongovernmental sectors who are interested in improving access and use of medicines for sick children. It is relevant at all levels of the health system—
- At the central level, for example, in the divisions of child health of the Ministry of Health, the national office of a mission health program, or the national health policy unit of a donor organization
- At the regional level, such as a provincial or district health office
- At the community level, for example, a nongovernmental organization or a community health board
- In health or educational institutions, for example, health facilities, pharmacies, private clinics, or schools
Unique features of the Guide
The guide provides a wealth of intervention examples and additional resources to help users gain an in-depth understanding of key issues related to problem assessment and the development and evaluation of interventions. While the examples are specific to common child illnesses, they will apply to all areas of child or adult health that require the use of medicines.
Every chapter has been developed to stand alone, so that users can start at the chapter of immediate relevance to their working group. Three fictional scenarios are used as a common thread throughout the guide to illustrate key points from each chapter and to help users relate recommendations to their local context. The scenarios include—
- A national team working centrally in the ministry of health to implement and evaluate IMCI or other national child health interventions
- A national malaria program developing a strategy to expand the use of rapid diagnostic testing to encourage appropriate antimalarial treatment for children with confirmed malaria
- A district team starting to implement integrated community case management for children with pneumonia, diarrhea, and malaria
The guide is practical and action-oriented. It does not provide in-depth instructions in quantitative or qualitative research methods or communicate step-by-step details about designing and implementing interventions. Instead, it refers users to additional resources that address these issues in depth.
The guide is available in different versions: one with all supporting documents contained in a packet together with the guide and a lighter version without the supporting documents, but with internal links to navigate the document and hyperlinks to documents available on the web. Both are available on the SIAPS website, or by request on USB or in print form.
[i] Initially published in 2008 as Improving Community Use of Medicines in the Management of Child Illness: A Guide to Developing Interventions under the RPM Plus program, the Guide was updated in 2015 under the SIAPS program. It was written and revised by members of the Drug Policy Research Group of Harvard Medical School and Harvard Pilgrim Health Care Institute in collaboration with staff from RPM Plus (2008) and SIAPS (2015).

